 
Latest Explore all the latest news and information on Physics World.These quantum numbers start from n=1, 2, 3, …. The energy levels are represented by an integer known as the Quantum number.The lowest energy level of an electron is called the Ground state.An electron moves from lower to higher states by gaining the energy and moves from higher energy states to lower energy states by losing energy.The electrons revolve in their orbits without losing energy.Bohr utilized the concept of quantisation and put forward the following points in his theory.Later, because of a few issues like the stability of the nucleus Bohr’s model was adopted.Īlso, read more about Adsorption, here.It states that all the positive charge is tightly compressed and concentrated at the centre of an atom and all electrons will be revolving around them as planets revolve around the sun.Most alpha particles are passed through gold foil un-deviated which lets us understand that all the positive charge is concentrated tightly at the centre of the atom and this agreement supported the hypothesis of the nucleus which was proposed by Rutherford’s Nuclear model. If this is true most alpha particles should be scattered because of the repulsive force between the positively charged nucleus and alpha particles. The positive charge of the atom is uniformly distributed throughout the volume. Through this experiment, we concluded that the model J.J Thomson had suggested can’t be correct because according to his model the atom is not hollow.He stated that electrons revolve around the nucleus in a well-defined path called “orbit”. 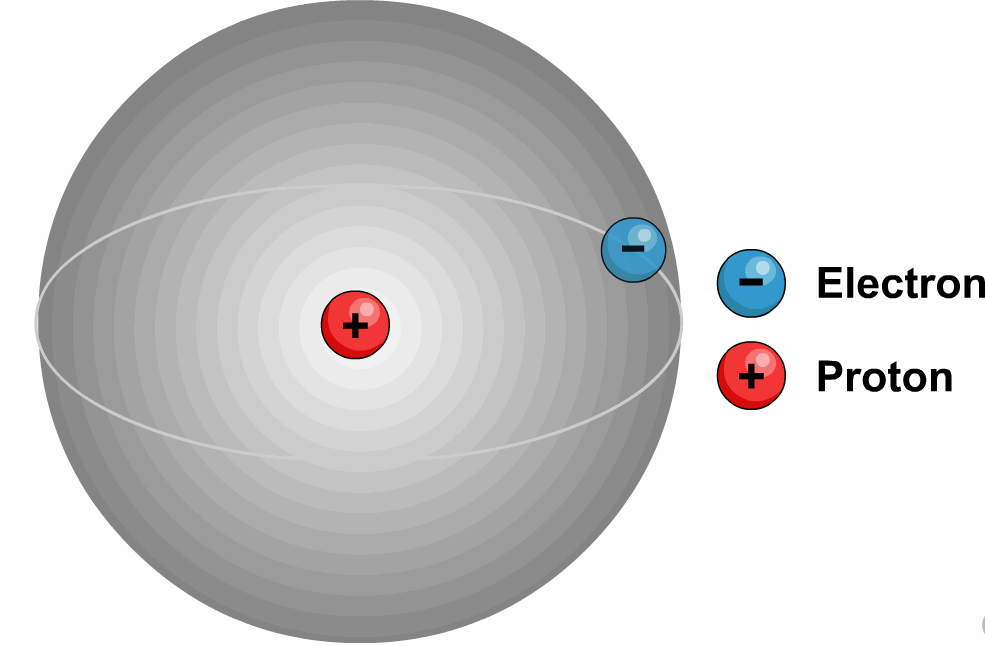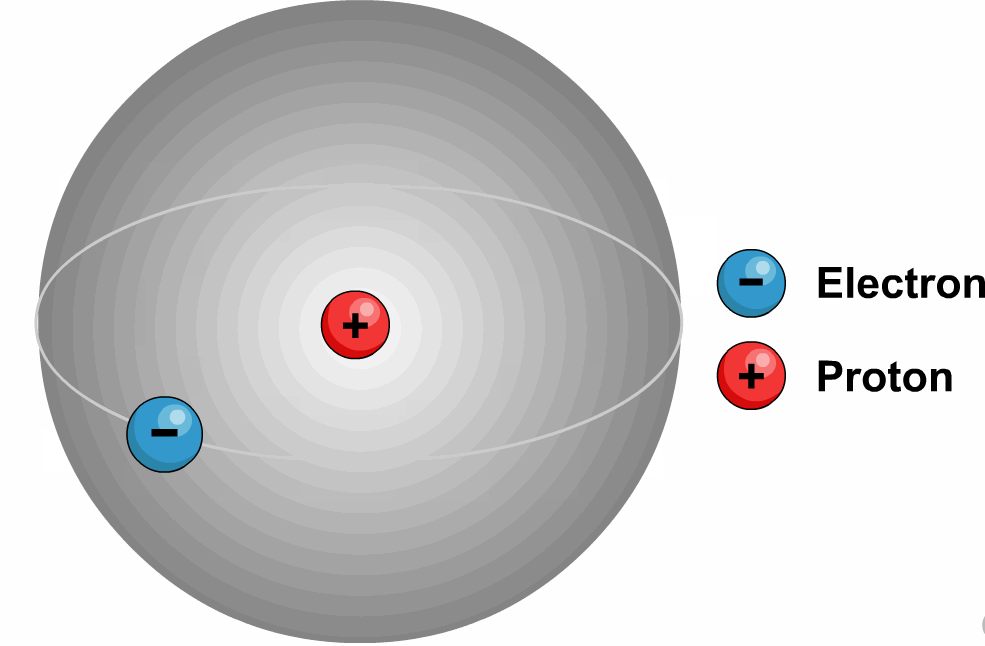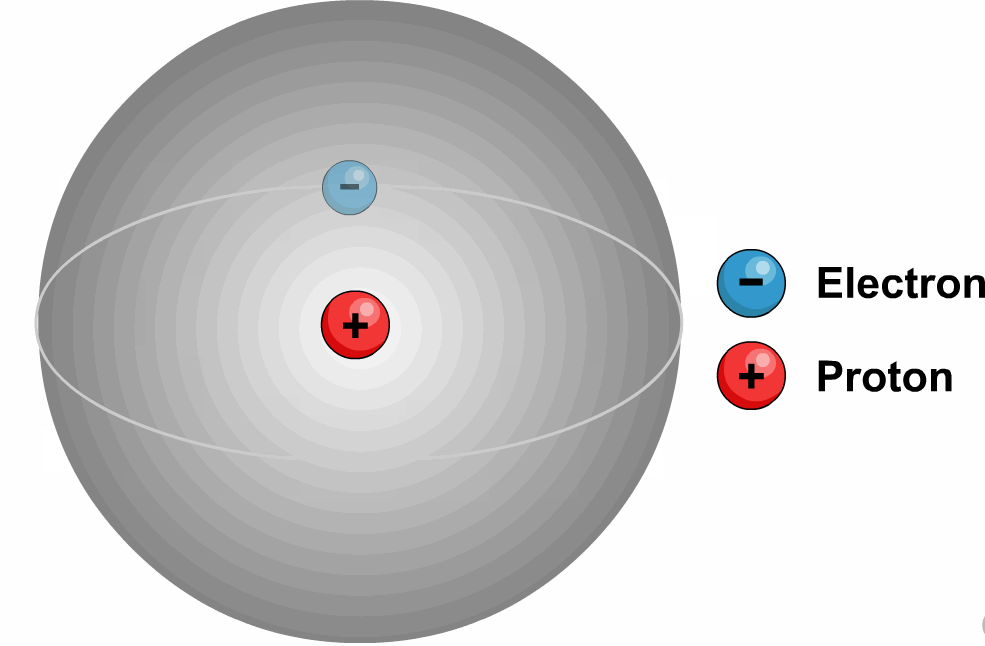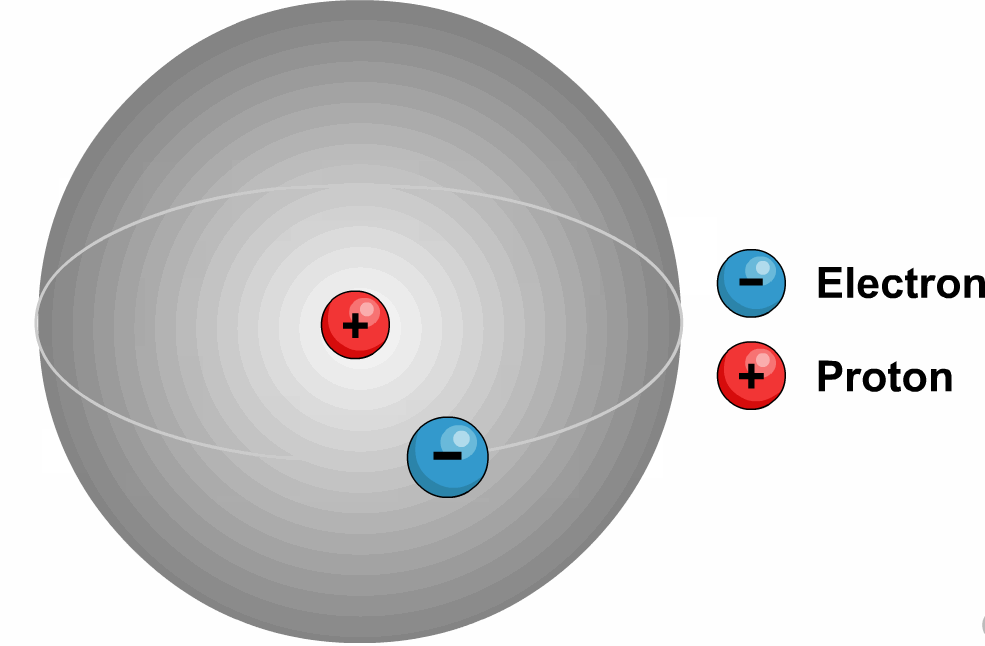 
The theory says that the size of the nucleus is very small as compared to the size of the atom.On the basis of this experiment, Rutherford stated that there is a positively charged spherical centre in an atom called the nucleus, and nearly all the mass of an atom (having a radius 10-10m) is packed in the nucleus (having a radius of 10–15 m).Some α–particles were deflected by small angles, and some were deflected by nearly 180°.He observed that most of the α– particles passed through the gold foil without deflecting.Rutherford used α–particle scattering experiment on a gold sheet.Know all about Solutions, their Components, Types, Properties here. It also failed to explain the reasons for the stability of an atom. 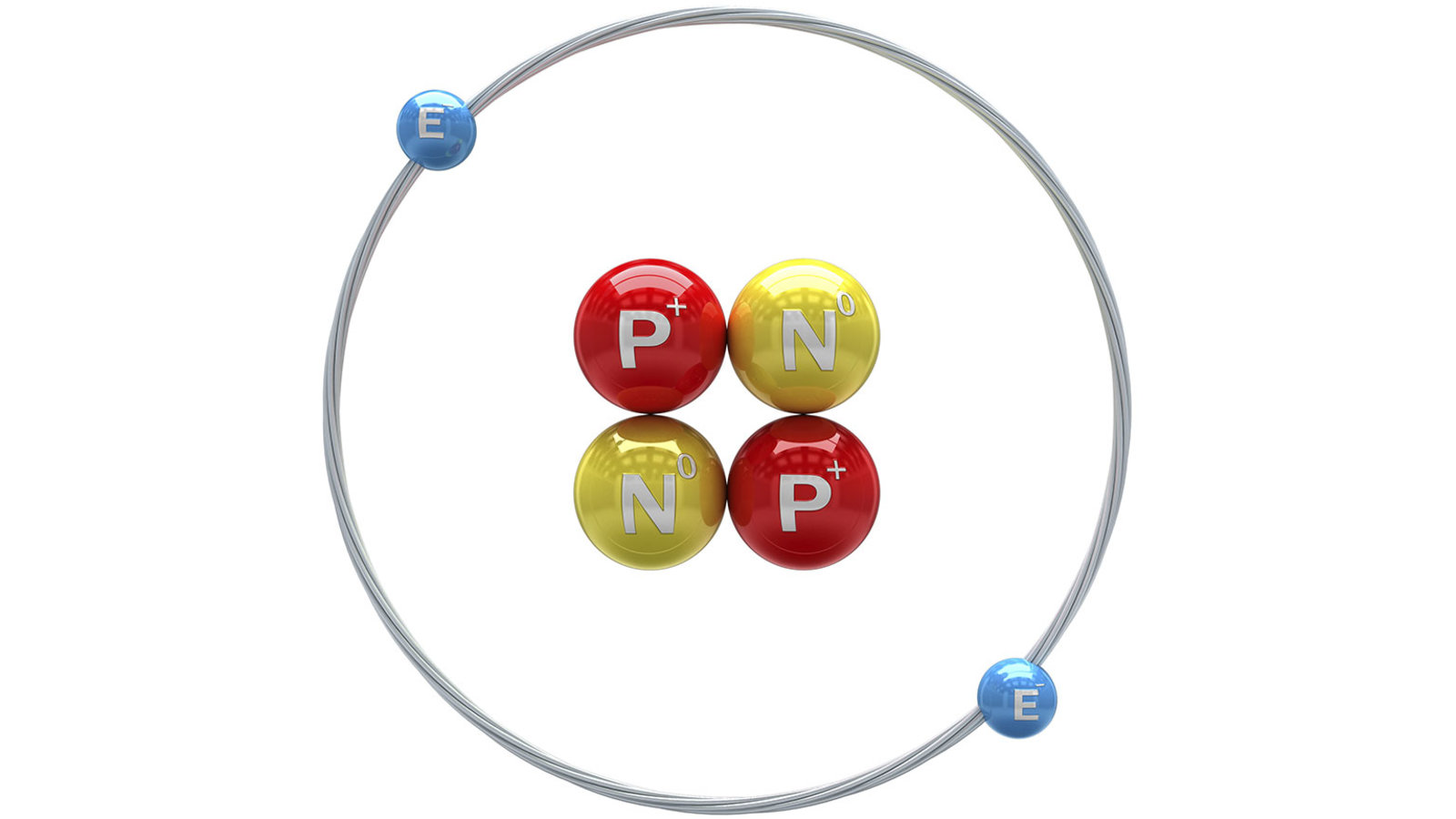
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
